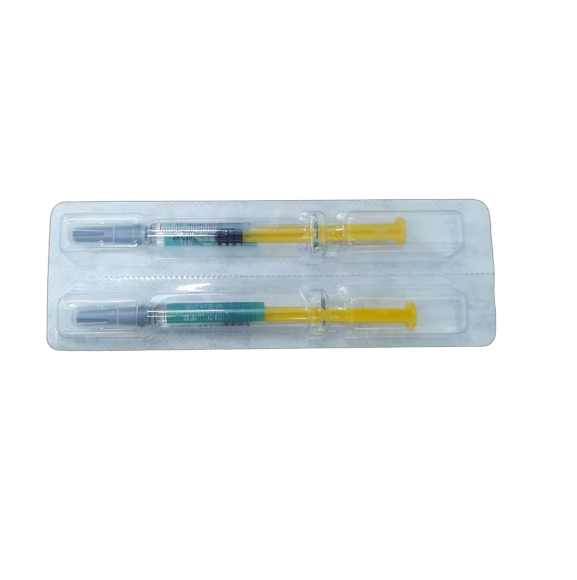
WAMIN – China exporter, global supplier & GMP manufacturer of Nadroparin Calcium Injection 0.3ml:2850 IU & 0.6ml:5700 IU
Introduction of WAMIN’s Nadroparin Calcium Injection
Nadroparin Calcium Injection is a kind of anticoagulant drugs. It is one of the most common used low molecular weight heparins used for the prophylaxis of thrombotic events and deep vein thrombosis, and prevent unstable angina and non-Q-wave myocardial infarction. WAMIN is Nadroparin Calcium Injection supplier in China, here we do a briefly introduction of our product.
- Product Name: Nadroparin Calcium Injection
- Strength:0.3ml: 2850 AXa IU; 0.4ml: 3800 AXa IU; 0.6ml: 5700 AXa IU; 0.8ml: 7600 AXa IU
- Standard: EP/CP
- Regular Packing:2 PFS/box, 200 box/carton
- Gross weight (per carton):7.2 kgs
- Net Weight (per carton): 6.0 kgs
- Measurement(per carton): 33cm*26cm*21cm
- Shelf life: 3 years
- Storage: Store in cool and dry place, protect from light, keep out of the sight and reach of children. Store below 25°C but do not freeze.
Nadroparin Calcium Injection Therapeutic Indications and Dosage
- In surgery, for the prevention of venous thromboembolic disease in cases of moderate or high risk of venous thrombosis.
- Treatment of deep vein thrombosis that has developed.
- Combined with aspirin for the treatment of unstable angina and non-Q-wave myocardial infarction in the acute phase.
- Prevention of blood clot formation in extracorporeal circulation in hemodialysis.
Nadroparin Calcium Injection Side Effects
Adverse reactions are listed below according to system organ class and frequency of occurrence
Adverse reactions are classified according to frequency using the following conventions: very common ≥1/10, common ≥1/100 and <1/10, uncommon ≥1/1000 and <1/100, rare ≥1/10000 and < 1/1000, very rare <1/10000.
Blood and lymphatic system disorders
- Very common: Haemorrhagic manifestations at various sites, more frequent in patients with other risk factors
- Rare: Thrombocytopenia (including heparin-induced thrombocytopenia), thrombocytosis
- Very rare: Hypereosinophilia, reversible after treatment discontinuation.
Immune system disorders
- Very rare: Hypersensitivity reactions (including angioedema and skin reactions), anaphylactoid reactions.
Metabolism and nutrition disorders
- Very rare: Reversible hyperkalemia associated with heparin-induced aldosterone suppression, especially in those with comorbid risk factors
Hepato-biliary disorders
- Common: Raised transaminases, usually transient
- Reproductive system and breast disorders
- Very rare: Priapism
Skin and subcutaneous tissue disorders
- Rare: Rash, urticaria, erythema, pruritus
- Very rare: Skin necrosis (usually at the injection site)
Systemic disorders and administration site conditions
- Very common: Small hematoma at the injection site.
In some cases, the emergence of firm nodules, which do not indicate an encystment of the heparin may be noted. These nodules usually disappear after a few days.
- Common: Injection site reactions
- Rare: Calcinosis at injection site.
Calcinosis is more frequent in patients with abnormal calcium and phosphorus deposition, such as in some cases of chronic renal failure.
Warnings
- Spinal or epidural hematoma. When performing spinal anesthesia (spinal anesthesia and epidural anesthesia) or spinal canal puncture, it should be noted that patients who use low molecular weight heparin or heparin-like substances to prevent thrombotic complications may cause spinal or epidural hematoma, leading to Long-term or even permanent paralysis. Epidural catheter placement or repeated epidural puncture, combined with the use of drugs that affect hemostatic function, such as non-steroidal anti-inflammatory drugs (NSAIDs), platelet coagulation agents, or other anticoagulant drugs, may lead to a higher incidence of hematoma. In this case, the patient should be monitored for symptoms and signs of nerve damage, and if it is found that the nerve may be damaged, it should be treated urgently. Physicians should weigh the pros and cons of intraspinal intervention (anesthesia or puncture) in such patients.
By WAMIN – Nadroparin Calcium Injection GMP manufacturer in China